1. HAMETAN cream
2. QUALITATIVE AND QUANTITATIVE COMPOSITION
Active ingredient:
Witch hazel virginia (potency 12mg / 100g) 5.35 g / 100 g Excipients:
Cetyl alcohol 5.00 g / 100 g Sorbic acid 0.15 g / 100 g
For excipients, see 6.1.
3. PHARMACEUTICAL FORM
Cream
It is a white, homogeneous and smooth cream. 4. CLINICAL FEATURES
4.1. Therapeutic indications HAMETAN;
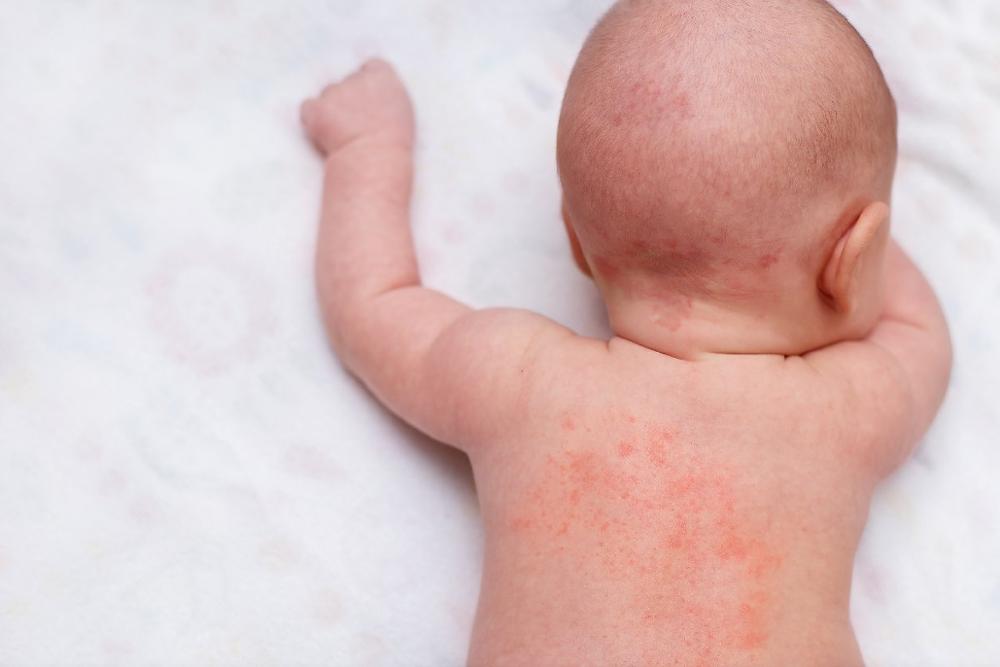
– In the diaper rash of infants and young children
– 1st and 2nd degree burns / sun burns
– In superficial skin lesions such as scratches and cuts
– In the care and protection of nipple cracks in nursing mothers
– Used for the care and protection of dried, cracked, split skin [including the skin of older people].
4.2. Posology and method of application
Posology / application frequency and duration:
HAMET that; It is applied to the lesioned area several times a day. There is no time limit for HAMETAN use. Method of Application:
HAMETAN is applied in a thin layer to the lesioned area of the skin.
If necessary, the lesioned area can be covered with gauze after application to the skin.
one
HAMETAN is applied to the nipples in the care of the nipples in nursing mothers after each breastfeeding.
Additional information on special populations: Kidney / Liver failure: Not reported. Pediatric population: Not reported. Geriatric population: Not reported.
4.3. contraindications
HAMETAN has no known contraindications.
However, as a general precaution, it should not be used in people who are hypersensitive to any of the ingredients in its composition.
4.4. Special use warnings and precautions
HAMETAN should not be in contact with the eyes. In case of contact with the eyes, it is recommended to wash the eyes with plenty of water.
If existing complaints persist or increase in the patient, a doctor should be consulted.
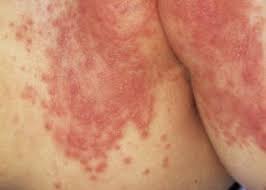
Due to its cetyl alcohol and sorbic acid content, it may cause local skin reactions (eg contact dermatitis).
4.5. Interactions with other medicinal products and other forms of interaction
No interaction has been reported.
4.6. Pregnancy and lactation
General advice
Pregnancy category has not been reported.
Women with childbearing potential / Contraception (Contraception)
Not applicable.
Gestation period
Clinical data on exposure to Hamamelis virginiana during pregnancy are not available.
Studies on animals do not show direct or indirect harmful effects related to fetal development.
Caution should be exercised when giving to pregnant women.
2nd
Lactation period
It is not known whether Hamamelis virginiana is excreted in human milk. The excretion of Hamamelis virginiana with milk has not been studied in animals. When deciding whether to stop breastfeeding or to stop HAMETAN treatment, the benefit of breastfeeding for the child and the benefit of HAMETAN treatment for the nursing mother should be taken into consideration.
Reproductive ability / Fertility
Not applicable.
4.7. Effects on the ability to drive and use machines
No effect of HAMETAN on vehicle and machine use has been reported.
8.4. Undesirable effects
There are no side effects reported to date.
4.9. Overdose and treatment
Not applicable.
5. PHARMACOLOGICAL FEATURES
5.1. Pharmacodynamic properties
Pharmacotherapeutic Group: Other Dermatologicals ATC Code: D11AX
Hamamelis virginiana aqueous distillate in the composition of HAMETAN;
has local hemostatic and anti-inflammatory effects.
With an astringent effect; Following skin injuries, it provides tissue collapsing and thickener effect by providing proteins to collapse in the area in question.
With its local hemostatic effect; Following the skin injuries, it shortens the bleeding time in the area in question, and speeds up the coagulation process.
With its anti-inflammatory effect; relieves inflammation symptoms and signs on the skin.
5.2. Pharmacokinetic properties General properties
Absorbation:
HAMETAN is an oil-in-water emulsion [containing 12% oil].
Thanks to the liposomes in its content; It has been reported that absorption by the skin is facilitated and reduces transepidermal moisture loss.
There is no information that it has a systemic effect.
astringent,
3
Distribution:
Not applicable. Biotransformation: Not reported.
Elimination:
Not applicable.
5.3. Preclinical safety data Not reported.
6. PHARMACEUTICAL FEATURES 6.1. List of excipients
Stearic acid
Cetyl alcohol
Polyoxyethylene-5-glycerin stearate (Arlatone 983 S) 85% Glycerol (glycerin)
Liquid paraffin (vaseline liquid)
Isopropyl myristate
Sorbic acid
Phosal 50 SA
Oxynex LM – Merck AG (Oksineks LM) Disodium edetate (EDTA disodium)
N-Sodium hydroxide solution (sodium hydroxide) Pure water
6.2. Incompatibilities
Not applicable.
6.3. Shelf life
24 months.
6.4. Special precautions for storage
Should be stored at room temperatures below 250C.




























There are no reviews yet.